
Collagen is the most abundant protein in the human body and provides the foundational structural framework of the extracellular matrix (ECM). Individual collagen polypeptide chains twist together to form a highly conserved triple-helical fibrillar structure—an ancient molecular architecture that enabled multicellularity and complex tissue evolution (1). In the cornea, this helical fibrillar architecture not only confers tensile strength but also forms the scaffold that directs epithelial adhesion, migration, integrin signaling, and cytoskeletal organization (2-5).
The corneal epithelium renews continuously through the coordinated activity of limbal epithelial stem cells, their transient amplifying progeny, and the basal epithelial cell layer, all of which rely on a continuously intact basement-membrane collagen substrate to support differentiation, migration, and wound healing. In dry eye disease, epithelial proliferation is paradoxically increased rather than decreased, representing a compensatory response to accelerated cell loss and premature desquamation of superficial cells (6). Disruption of the underlying collagen foundation compromises this epithelial homeostasis and contributes to the recurrent surface breakdown observed in dry eye disease.
Collagen architecture
The stromal lamellae contain a highly ordered, periodic array of collagen fibrils with uniform diameter and spacing. This lattice produces destructive interference of scattered light, enabling corneal transparency. Proteoglycans and regulated stromal hydration maintain this micro-architecture and preserve optical quality (7).
Collagen in Bowman's layer and the anterior stroma provides the anchoring substrate for corneal epithelial cells. Engagement of epithelial integrins (particularly α2β1, α3β1, and α6β4) with this collagen-rich matrix activates PI3K, ERK, and Rho pathways that orchestrate cytoskeletal organization, tight-junction function, and directed epithelial migration (2-5, 8). Although fibrillar collagen is not itself a structural component of tight junctions, intact collagen is indispensable for the integrin-mediated signaling that organizes the perijunctional actin ring and maintains barrier integrity. When collagen becomes fragmented, impaired integrin engagement disrupts Rho- and PI3K-dependent cytoskeletal dynamics, leading to defective tight-junction formation and increased epithelial permeability.
How dry eye disrupts collagen and epithelial homeostasis
Desiccating stress destabilizes the tear film and upregulates inflammatory mediators, including MMP-9 and IL-6.9 Experimental studies in humans using a controlled adverse environment (CAE) chamber have shown that as little as 90 minutes of exposure to desiccating stress can significantly destabilize the tear film, causing cell apoptosis and sharp increases in tear levels of MMP-9 and IL-6.10,11 MMP-9, which is elevated in tears of dry eye patients, preferentially degrades basement-membrane collagen (particularly type IV collagen and laminin) and denatured collagen fragments, undermining the structural foundation required for epithelial stability (12, 13).
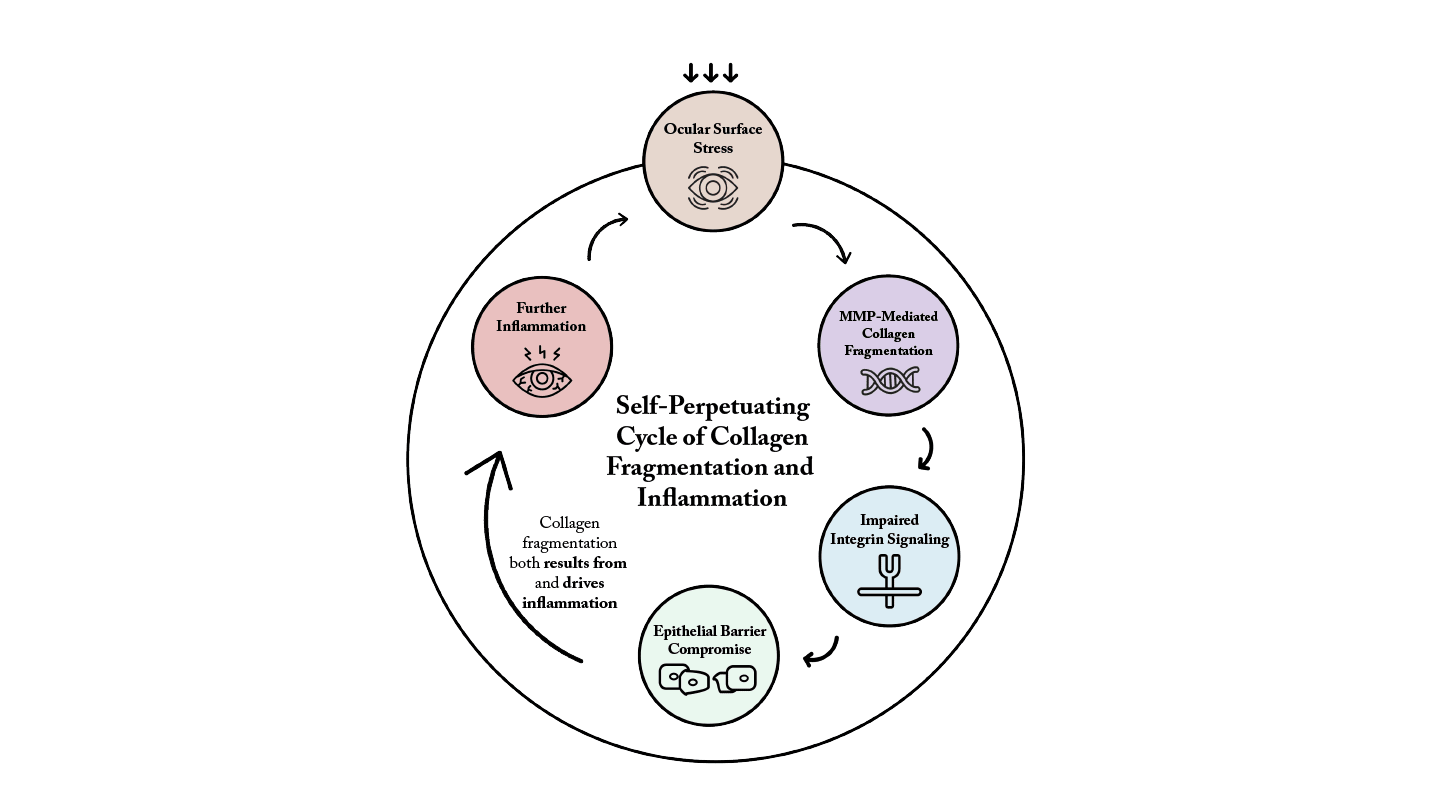
Fragmented collagen helices act as damage-associated molecular patterns (DAMPs), activating TLR-2, TLR-4, chemokine receptors, and inflammatory integrins (14, 15). Fragmented collagen not only reflects tissue injury but also acts as a pro-inflammatory ligand, directly promoting inflammation by altering receptor-ligand interactions at the cell surface and facilitating immune cell motility. This leads to increased chemokine and cytokine production and immune cell infiltration. Collagen fragmentation therefore both results from, and drives, inflammation (Figure 1), creating a self-amplifying cycle that disrupts integrin signaling, impairs cytoskeletal organization and weakens epithelial barrier function through accelerated desquamation of occludin-bearing apical cells (16).
The optical consequences of superficial punctate keratitis (SPK)
SPK generates micro-irregularities in the epithelial surface that disrupt the smooth refractive interface formed by the epithelium and tear film. These irregularities increase forward scatter, degrade contrast sensitivity, and elevate higher-order aberrations (particularly coma and trefoil), producing glare, halos, and fluctuating clarity (17-19). The severity of epithelial damage at the central cornea correlates significantly with both visual maintenance ratio and higher-order aberrations, which dynamically worsen over time after each blink in dry eye patients (17, 20). Many patients experience momentary crisp vision immediately after a blink, followed by rapid deterioration as the tear film destabilizes over SPK lesions.
Central epithelial irregularity impairs the reliability of keratometry and corneal topography, producing inconsistent measurements that can affect IOL power calculations in patients preparing for cataract surgery (21). Although axial length remains unaffected, SPK introduces variability into corneal power measurements. Mechanical factors such as lid–cornea friction in mucin-deficient states and hyperosmolarity-induced apoptosis further shape the staining pattern, with reduced corneal nerve density and impaired corneal sensitivity contributing to altered ocular surface homeostasis in dry eye disease (22, 23).
Collagen mimetics as targeted structural-reparative therapy
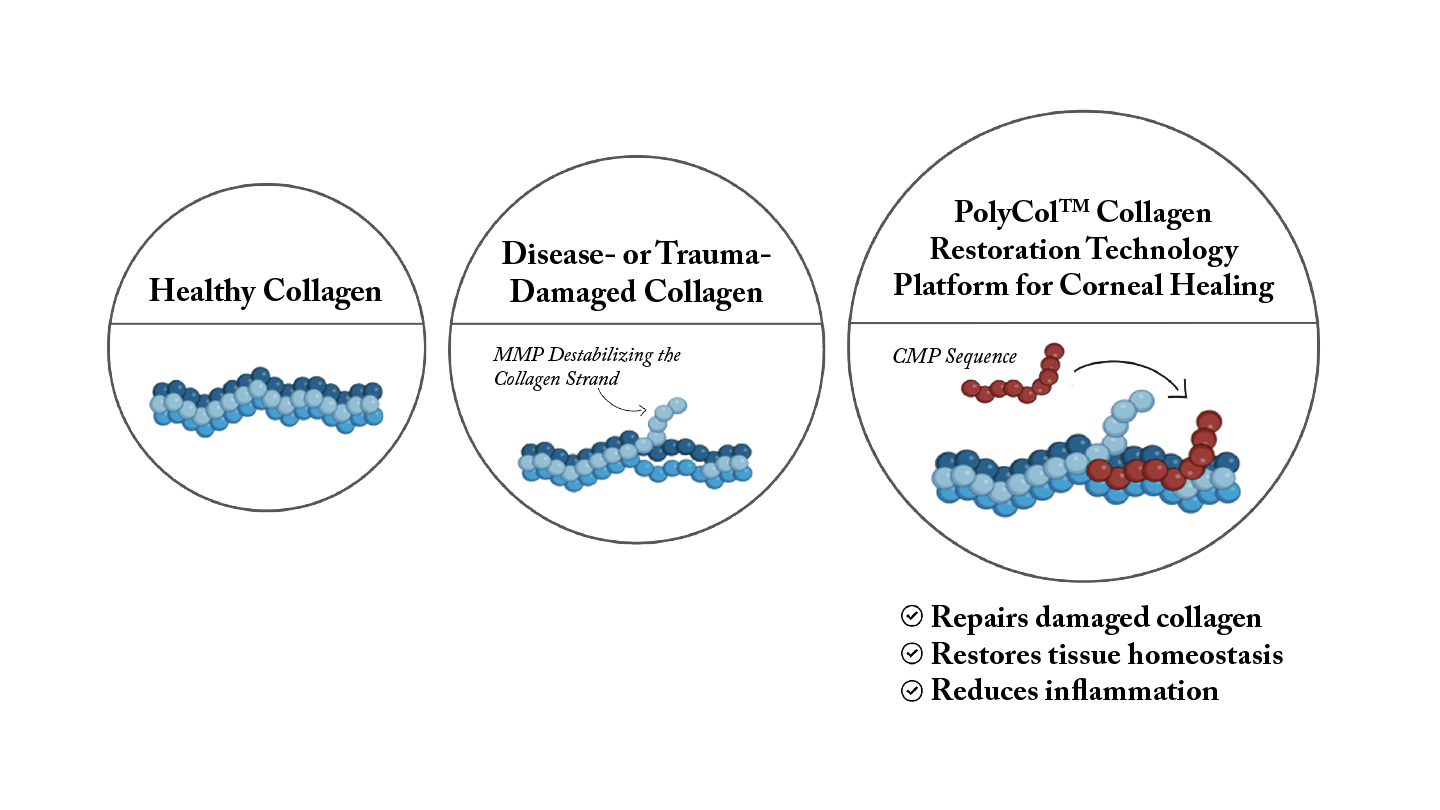
Collagen mimetic peptides (CMPs) anneal selectively to fragmented collagen helices—without binding intact fibrils—and therefore localize specifically to sites of ECM injury (Figure 2) (24, 25). By bridging damaged collagen fragments, CMPs promote reconstitution of the triple-helical structure, restoring ECM continuity. Repaired collagen scaffolding enables integrins to reengage physiologically with native ligands, reactivating PI3K, ERK, and Rho signaling required for cytoskeletal remodeling, epithelial migration, and barrier reformation (26-28).
It is important to note that when collagen in the eye becomes fragmented or damaged, it typically is not repaired; rather, it is slowly replaced through a natural remodeling process. With age, that collagen remodeling rate slows down, which can be a contributor to certain effects arising from chronic disease. CMPs act as molecular building blocks that bind to and repair fragmented collagen helices in the corneal ECM, directly repairing damaged segments and promoting collagen re-assembly into a functional network—a fundamentally different approach from the slow natural replacement process or from physical collagen shields that function as temporary external dressings.
Clinical evidence supporting collagen mimetics: The ST-100 phase 2 trial
ST-100 (vezocolmitide, Stuart Therapeutics), a topical collagen mimetic peptide consisting of repeated Pro-Pro-Gly tripeptide sequences, was evaluated in a multicenter, randomized, double-masked Phase 2 clinical trial (29). Twice-daily ST-100 produced clinically meaningful reductions in corneal fluorescein staining, with the greatest improvements in the central and inferior cornea. The treatment also demonstrated statistically significant increases in Schirmer test scores compared to vehicle, with a Schirmer responder rate (change from baseline ≥10 mm) of 12.2% for high-dose ST-100 versus 0.0% for vehicle.
Improvement in staining was observed as early as 48 hours in some participants. Rapid restoration of epithelial integrity is clinically meaningful—particularly for patients preparing for refractive cataract surgery or keratorefractive procedures, where ocular surface instability undermines keratometric and refractive consistency and delays surgical planning. The treatment was well-tolerated with adverse events comparable to vehicle controls and no significant issues with burning or stinging on instillation.
In preclinical studies, mice subjected to desiccating air flow and topical application of 1% atropine solution to simulate dry eye with corneal nerve damage showed that CMP treatment reduced expression of inflammatory markers and improved corneal epithelium integrity, tear film production, and corneal sensitivity (30). Compellingly, ST-100 was able to expand the sub-basal nerve plexus of the cornea, which helps maintain the homeostatic function of the ocular surface.
Why collagen mimetics represent a novel disease-modifying therapy for dry eye
Dry eye disease is a multifactorial condition involving tear film instability, inflammation, and neurosensory dysfunction. While existing therapies target inflammatory pathways—such as cyclosporine (T-cell immunomodulation) and lifitegrast (LFA-1 antagonism)—collagen degradation appears to be an upstream structural driver that both results from and perpetuates inflammation (15, 31).
By annealing to fragmented collagen and restoring triple-helical structure, CMPs may interrupt the positive feedback loop of inflammation → MMP-9 upregulation → collagen fragmentation → DAMP signaling → further inflammation. Through structural repair, CMPs normalize integrin engagement, stabilize cytoskeletal dynamics, promote epithelial renewal, and reduce susceptibility to further enzymatic degradation. Additionally, CMPs have demonstrated direct anti-inflammatory effects, reducing secretion of pro-inflammatory cytokines (IL-6, IL-8) and reactive oxygen species in preclinical models (28, 32). Recent studies also show that CMPs repair the corneal sub-basal nerve plexus in dry eye models, reducing nerve fragmentation and expanding neuronal coverage, which may contribute to improved corneal sensitivity and function (30).
This novel mechanism of action positions CMPs as both a potential first-line therapy with rapid onset of epithelial repair and as a complementary treatment that may work synergistically with existing anti-inflammatory therapies. By addressing the structural foundation that underlies epithelial barrier dysfunction, CMPs offer a mechanistically distinct approach that targets the root cause of dry eye pathology while potentially enhancing the efficacy of immunomodulatory agents. Importantly, CMP therapy would likely be agnostic to the type of dry eye—whether primarily aqueous-deficient, evaporative secondary to meibomian gland dysfunction, or a mixed form—as all subtypes involve collagen degradation as a common pathway in the inflammatory cascade.
Conclusion
Collagen degradation is a central, under-recognized structural pathology in dry eye disease. Collagen mimetic peptides restore ECM integrity, normalize integrin signaling, and represent a novel regenerative therapeutic approach that could serve as both a first-line therapy and complement existing disease-modifying treatments for dry eye. None of our current dry eye treatments offer regenerative benefits; CMPs have the potential to be truly disease-modifying for dry eye by repairing the structural foundation of the corneal surface.
References
- AL Fidler et al., “The triple helix of collagens—an ancient protein structure that enabled animal multicellularity and tissue evolution,” J Cell Sci, 131, jcs203950 (2018).
- MA Stepp, “Corneal integrins and their functions,” Exp Eye Res, 83, 3 (2006).
- AV Ljubimov, M Saghizadeh, “Progress in corneal wound healing,” Exp Eye Res, 133, 132 (2015).
- TB McKay et al., “Integrin: Basement membrane adhesion by corneal epithelial and endothelial cells,” Exp Eye Res, 198, 108138 (2020).
- A Manninen, “Epithelial polarity—generating and integrating signals from the ECM with integrins,” Exp Cell Res, 334, 337 (2015).
- C Fabiani et al., “Corneal epithelial proliferation and thickness in a mouse model of dry eye,” Exp Eye Res, 89, 166 (2009).
- KM Meek, C Knupp, “Corneal structure and transparency,” Prog Retin Eye Res, 49, 1 (2015).
- H Guo et al., “Integrin alpha v beta 6 contributes to maintaining corneal epithelial barrier function,” Cell Biol Int, 37, 593 (2013).
- AJ Bron et al., “TFOS DEWS II Pathophysiology Report,” Ocul Surf, 15, 438 (2017).
- I Fernández et al., “Response profiles to a controlled adverse desiccating environment based on clinical and tear molecule changes,” Ocul Surf, 17, 502 (2019).
- A López-Miguel et al., “Dry eye exacerbation in patients exposed to desiccating stress under controlled environmental conditions,” Am J Ophthalmol, 157, 788 (2014).
- R Sambursky et al., “Sensitivity and specificity of a point-of-care matrix metalloproteinase 9 immunoassay for diagnosing inflammation related to dry eye,” JAMA Ophthalmol, 131, 24 (2013).
- A Shoari et al., “Inhibition of matrix metalloproteinase-9 for the treatment of dry eye syndrome; a review study,” Exp Eye Res, 205, 108523 (2021).
- TL Adair-Kirk, RM Senior, “Fragments of extracellular matrix as mediators of inflammation,” Int J Biochem Cell Biol, 40, 1101 (2008).
- RO Baratta et al., “Corneal collagen as a potential therapeutic target in dry eye disease,” Surv Ophthalmol, 67, 60 (2022).
- SC Pflugfelder et al., “Matrix metalloproteinase-9 knockout confers resistance to corneal epithelial barrier disruption in experimental dry eye,” Am J Pathol, 166, 61 (2005).
- M Kaido et al., “Corneal fluorescein staining correlates with visual function in dry eye patients,” Invest Ophthalmol Vis Sci, 52, 9516 (2011).
- S Koh, “Irregular astigmatism and higher-order aberrations in eyes with dry eye disease,” Invest Ophthalmol Vis Sci, 59, DES36 (2018).
- S Koh et al., “The effect of ocular surface regularity on contrast sensitivity and straylight in dry eye,” Invest Ophthalmol Vis Sci, 58, 2647 (2017).
- A Denoyer et al., “Tear film aberration dynamics and vision-related quality of life in patients with dry eye disease,” Ophthalmology, 119, 1811 (2012).
- M Abou Shousha et al., “Corneal epithelial thickness profile in dry-eye disease,” Eye (Lond), 34, 915 (2020).
- DR Korb et al., “Lid wiper epitheliopathy and dry eye symptoms,” Eye Contact Lens, 31, 2 (2005).
- A Labbé et al., “Corneal nerve structure and function in patients with non-Sjogren dry eye: Clinical correlations,” Invest Ophthalmol Vis Sci, 54, 5144 (2013).
- SM Yu et al., “Collagen mimetic peptides: Progress towards functional applications,” Adv Drug Deliv Rev, 113, 248 (2011).
- P Ratnatilaka Na Bhuket et al., “From collagen mimetics to collagen hybridization and back,” Acc Chem Res, 57, 1649 (2024).
- LK Wareham et al., “Collagen in the central nervous system: Contributions to neurodegeneration and promise as a therapeutic target,” Mol Neurodegener, 19, 11 (2024).
- GO Bou Ghanem et al., “Collagen mimetic peptides promote repair of MMP-1-damaged collagen in the rodent sclera and optic nerve head,” Int J Mol Sci, 24, 17031 (2023).
- M Ribeiro et al., “Collagen mimetic peptides promote adherence and migration of ARPE-19 cells while reducing inflammatory and oxidative stress,” Int J Mol Sci, 23, 7004 (2022).
- RO Baratta et al., “A phase 2 trial to test safety and efficacy of ST-100, a unique collagen mimetic peptide ophthalmic solution for dry eye disease,” Ophthalmol Sci, 4, 100451 (2023).
- LK Wareham et al., “Collagen mimetic peptide repair of the corneal nerve bed in a mouse model of dry eye disease,” Front Neurosci, 17, 1148950 (2023).
- SC Pflugfelder, CS de Paiva, “The pathophysiology of dry eye disease: What we know and future directions for research,” Ophthalmology, 124, S4 (2017).
- J Elango et al., “The molecular interaction of collagen with cell receptors for biological function,” Polymers, 14, 876 (2022).
