Despite current anti-VEGF therapies for wet age-related macular degeneration (AMD) and diabetic macular edema (DME), many patients still experience long-term vision loss (1, 2). Patients with wet AMD and DME are often undertreated over the long term, with high rates of treatment discontinuation (1, 2). Lower treatment intensity may help explain why real-world visual outcomes with anti-VEGF therapies fall short of those reported in clinical trials (1, 10). Undertreatment is driven in part by the high treatment burden associated with regular intravitreal injections (3, 4), with dosing intervals typically ranging from every four to 16 weeks (5-9).
Moreover, retinal diseases are multifactorial, involving angiogenesis, vascular permeability, inflammation, and other pathogenic mechanisms (11, 12). Thus, response to anti-VEGF monotherapy may be limited, as these agents only target angiogenesis and vascular permeability, with inflammation still playing a key role. Multiple studies demonstrate that fluid fluctuations with anti-VEGF treatment are associated with worse visual outcomes (13-16), potentially due to intermittent, pulsatile dosing that can permit recurrent exudative activity, retinal instability, and subsequent tissue damage. Therefore, there is an unmet need for more durable treatment options that maintain optimal visual and anatomic stability while also reducing treatment burden.
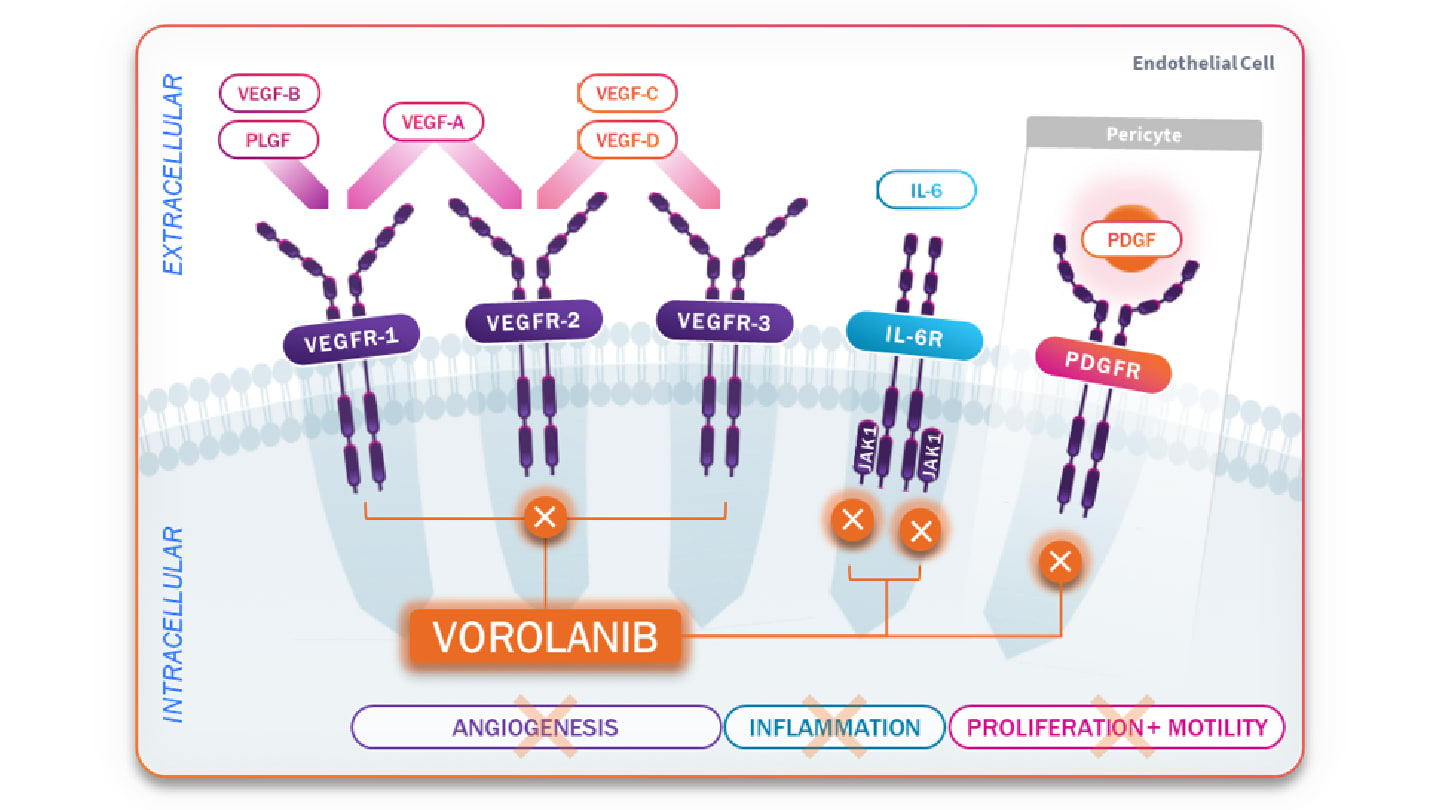
EYP-1901 (vorolanib intravitreal insert; DURAVYU™)* utilizes next-generation bioerodible Durasert E™ technology to provide sustained release of the tyrosine kinase inhibitor (TKI) vorolanib at therapeutic levels for at least six months after a single injection. Vorolanib offers a novel multi-mechanism of action, working intracellularly to suppress angiogenesis through the inhibition of all vascular endothelial growth factor receptors (VEGFRs) and platelet-derived growth factor receptor (PDGFR), along with suppression of inflammation via inhibition of interleukin-6 (IL-6) signaling (Figure 1) (17). EYP-1901 has been evaluated in a robust clinical trial program, including the proof-of-concept phase 1 DAVIO (NCT04747197) trial in wet AMD, phase 2 DAVIO 2 (NCT05381948) trial in wet AMD, and phase 2 VERONA (NCT06099184) trial in DME (18-20). Key findings from the DAVIO 2 and VERONA studies were featured in three presentations at the 49th Macula Society Annual Meeting.
DAVIO 2 enrolled 161 patients with previously-treated wet AMD who were randomized to receive a single 2 mg or 3 mg dose of EYP-1901 or aflibercept 2 mg every-8-weeks (q8W), following three loading doses of aflibercept 2 mg. All patients were eligible for supplemental aflibercept injections starting at Week 12, per prespecified vision and anatomical criteria (21).
Vision outcomes from DAVIO 2, including a post hoc time-in-range analysis, were presented by Dr. Rishi P. Singh. DAVIO 2 met its primary endpoint, with EYP-1901 2 mg and 3 mg both demonstrating statistical noninferiority to aflibercept 2 mg q8W on best-corrected visual acuity (BCVA) change from baseline averaged over Week 28/32. Over 85% of EYP-1901-treated patients had stable (BCVA change <5 letters) or improved (BCVA gain ≥5 letters) vision 6 months after a single dose (both dose levels), including >90% with EYP-1901 3 mg. Functional vision was sustained with a single EYP-1901 dose; time in range with EYP-1901 was similar across all thresholds compared to aflibercept 2 mg q8W. BCVA was maintained ≥69 letters, the minimum driving requirement in most US states, for the majority of time with EYP-1901 (EYP-1901 2 mg, 25 of 32 weeks; EYP-1901 3 mg, 28 of 32 weeks) (21).
Dr. Charles C. Wykoff presented findings from a DAVIO 2 post hoc analysis using AI segmentation of optical coherence tomography (OCT) images to evaluate retinal fluid dynamics. In addition to stable vision, patients treated with a single dose of EYP-1901 had stable central subfield thickness (CST) for 6 months after administration, with no sawtooth pattern as observed in the aflibercept 2 mg q8W arm. EYP-1901 at both doses controlled retinal fluid, particularly intraretinal fluid (IRF), comparable to aflibercept 2 mg q8W (22).
In DME, results from the VERONA trial were presented by Dr. Dilraj S. Grewal. VERONA enrolled 27 previously treated patients who received a single dose of aflibercept 2 mg followed by a single 1.3 mg or 2.7 mg dose of EYP-1901 or sham. The primary endpoint was met; both EYP-1901 dose levels achieved extended time-to-first supplemental injection versus aflibercept. Vision and anatomic improvements with EYP-1901 were seen as early as Week 4 (the first post-treatment visit) in the EYP-1901 arms and sustained throughout the trial (mean BCVA change from baseline: +7.1, +6.9, +7.3 letters; mean CST change from baseline: −75.9 μm, −71.1 μm, −43.7 μm, for EYP-1901 2.7 mg, 1.3 mg, and aflibercept arms, respectively, at Week 24). Greater reductions in mean macular leakage area (−3.1, −2.0, vs −0.7 mm2) and mean total macular volume (−0.60, −0.37, vs −0.16 mm3) were observed with EYP-1901 2.7 mg and 1.3 mg compared to aflibercept control at Week 24 (23).
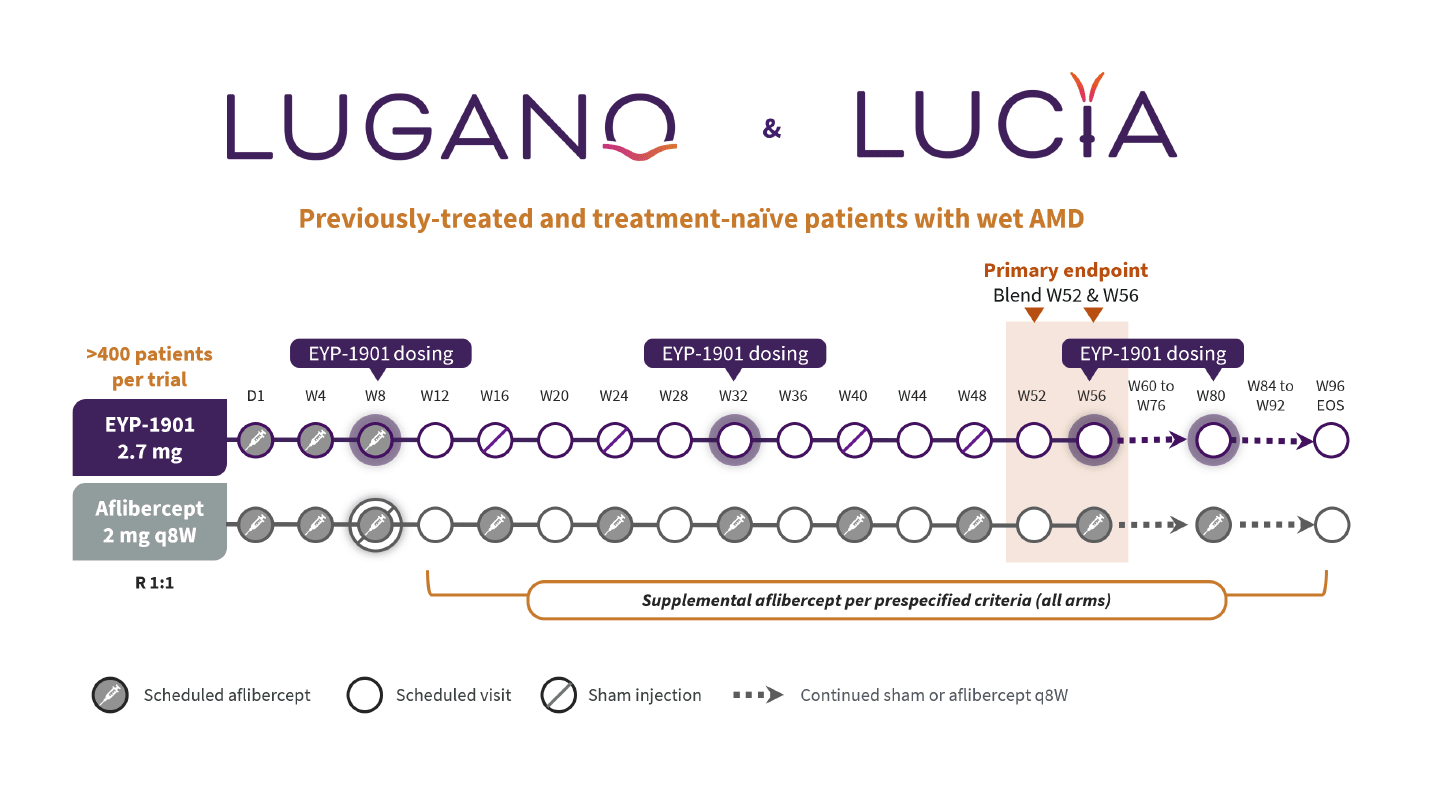
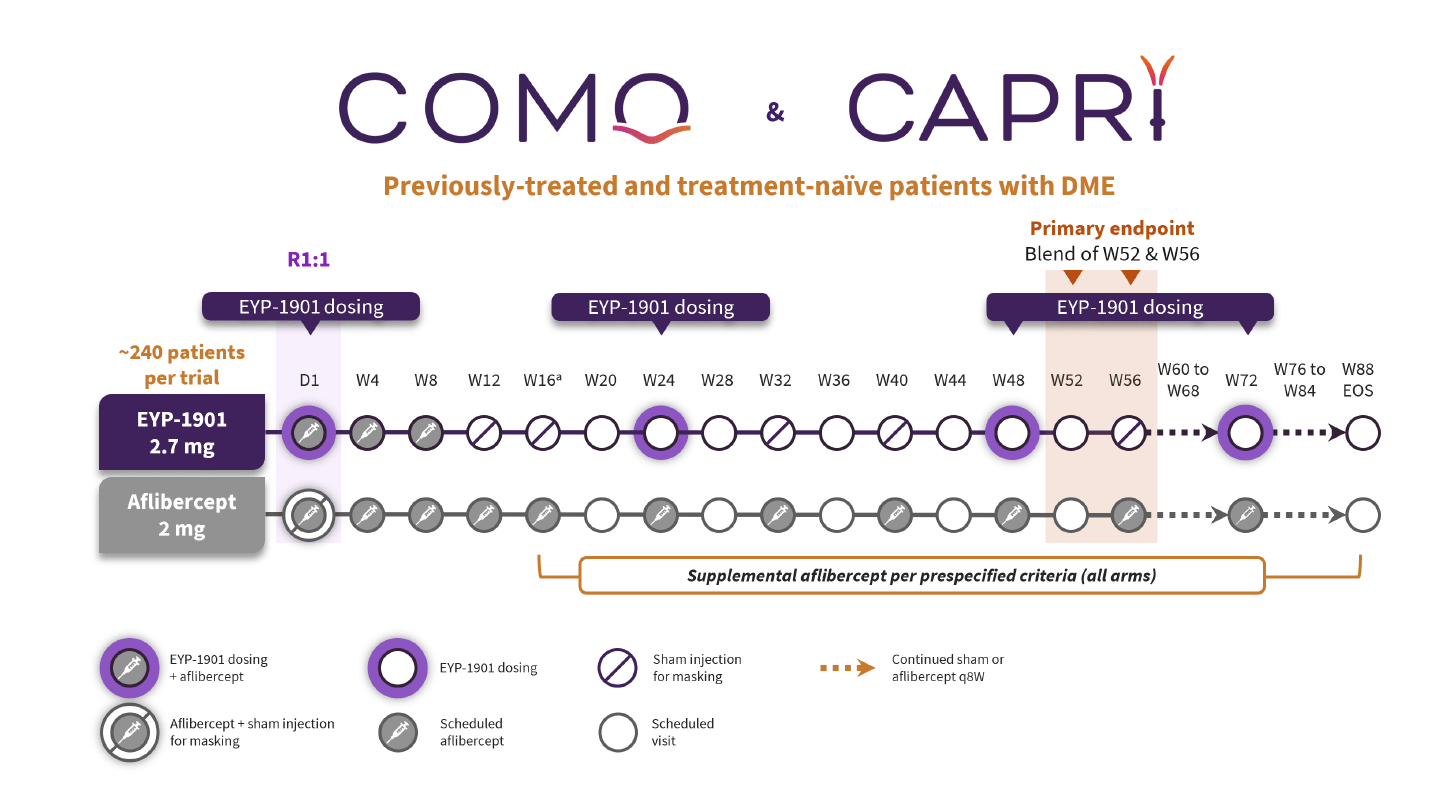
Across four completed clinical trials to date, EYP-1901 has demonstrated a favorable tolerability profile with no EYP-1901-related safety signals observed (17). Informed by the positive phase 2 trials, pivotal phase 3 trials evaluating EYP-1901 with six-month redosing versus on-label aflibercept 2 mg q8W are underway in both wet AMD and DME. The ongoing LUGANO (NCT06668064) and LUCIA (NCT06683742) trials enrolled 432 and 475 treatment-naïve and previously-treated patients with wet AMD, respectively (Figure 2). Topline data for LUGANO are anticipated mid-2026, with LUCIA to shortly follow (24, 25). Preparations for the pivotal COMO and CAPRI trials in DME are ongoing (ClinicalTrials.gov registration pending). Each trial will enroll approximately 240 treatment-naïve and previously-treated patients with DME (Figure 3), with first patient dosing anticipated first quarter of 2026 (26). A comprehensive, robust clinical trial program supports EYP-1901’s potential to improve long-term visual outcomes and reduce treatment burden in wet AMD and DME.
*DURAVYU™ has been conditionally accepted by the US FDA as the proprietary name for
EYP-1901 (vorolanib intravitreal insert). EYP-1901 is an investigational medicinal product and is not authorized for sale in any country at the time of this publication. FDA approval in the US and Marketing Authorization in any other country and the timeline for potential approval or authorization is uncertain.
References
- CC Wykoff et al., “Impact of Anti-VEGF Treatment and Patient Characteristics on Vision Outcomes in Neovascular Age-related Macular Degeneration: Up to 6-Year Analysis of the AAO IRIS® Registry,” Ophthalmol Sci, 4, 100421 (2024). PMID: 38312345.
- BL Kuo et al., “Long-term Treatment Patterns for Diabetic Macular Edema: Up to 6-Year Follow-up in the IRIS® Registry,” Ophthalmol Retina, 8, 1074 (2024). PMID: 38945672.
- TA Ciulla et al., “Longer-Term Anti-VEGF Therapy Outcomes in Neovascular Age-Related Macular Degeneration, Diabetic Macular Edema, and Vein Occlusion-Related Macular Edema: Clinical Outcomes in 130 247 Eyes,” Ophthalmol Retina, 6, 796 (2022). PMID: 35432109.
- A Loewenstein et al., “Global insights from patients, providers, and staff on challenges and solutions in managing neovascular age-related macular degeneration,” Ophthalmol Ther, 14, 211 (2025).
- Genentech, “LUCENTIS® (ranibizumab injection) Prescribing Information,” South San Francisco, CA (2024).
- Genentech, “VABYSMO® (faricimab-svoa) Prescribing Information,” South San Francisco, CA (2024).
- Regeneron Pharmaceuticals, “EYLEA® (aflibercept) Prescribing Information,” Tarrytown, NY (2024).
- Regeneron Pharmaceuticals, “EYLEA HD® (aflibercept) Prescribing Information,” Tarrytown, NY (2025).
- Novartis Pharmaceuticals Corporation, “BEOVU® (brolucizumab-dbll) Prescribing Information,” East Hanover, NJ (2024).
- TA Ciulla et al., “Visual acuity outcomes and anti-VEGF therapy intensity in diabetic macular oedema: a real-world analysis of 28 658 patient eyes,” Br J Ophthalmol, 105, 216 (2021). PMID: 32948563.
- M Fleckenstein et al., “Age-related macular degeneration: a review,” JAMA, 331, 147 (2024).
- S Vujosevic et al., “Role of inflammation in diabetic macular edema and neovascular age-related macular degeneration,” Surv Ophthalmol, 69, 870 (2024). PMID: 39123487.
- U Chakravarthy et al., “Impact of macular fluid volume fluctuations on visual acuity during anti-VEGF therapy in eyes with nAMD,” Eye (Lond), 35, 2983 (2021).
- PU Dugel et al., “Effect of retinal thickness variability on visual outcomes and fluid persistence in neovascular age-related macular degeneration: a post hoc analysis of the HAWK and HARRIER studies,” Retina, 42, 511 (2022).
- RN Evans et al., “Associations of variation in retinal thickness with visual acuity and anatomic outcomes in eyes with neovascular age-related macular degeneration lesions treated with anti–vascular endothelial growth factor agents,” JAMA Ophthalmol, 138, 1043 (2020).
- AW Zhou et al., “Macular thickness fluctuation as a biomarker: impact on visual acuity following antivascular endothelial growth factor therapy for diabetic macular edema,” J Vitreoretin Dis, 10, 83 (2026).
- JS Duker, “DURAVYU: Sustained-Release, Multi-MoA TKI with the Potential to Fulfill the Unmet Needs in DME and Wet AMD,” Presented at: Eyecelerator @ AAO; Denver, CO (2025). Accessed January 6, 2026.
- ClinicalTrials.gov, “First in Human Study to Evaluate the Safety and Tolerability of EYP-1901 in Patients With Wet Age Related Macular Degeneration (wAMD),” (2021). Accessed February 20, 2026.
- ClinicalTrials.gov, “Study of EYP-1901 in Subjects With Wet Age Related Macular Degeneration (wAMD) (DAVIO2),” (2022). Accessed February 20, 2026.
- ClinicalTrials.gov, “Study of EYP-1901 in Patients With Diabetic Macular Edema (DME) (VERONA),” (2023). Accessed February 20, 2026.
- RP Singh, “Visual Acuity Time-in-Range Analysis with Bioerodible EYP-1901 (vorolanib intravitreal insert) Versus Aflibercept in Previously-Treated Neovascular Age Related Macular Degeneration,” Presented at: 49th Macula Society Annual Meeting; San Diego, CA (2026).
- CC Wykoff, “Retinal Fluid Dynamics with the Sustained-release, Bioerodible Insert EYP-1901 (vorolanib intravitreal insert): Higher-order OCT Analytics Using AI Segmentation of Images from the DAVIO 2 Trial,” Presented at: 49th Macula Society Annual Meeting; San Diego, CA (2026).
- DS Grewal, “Visual, Anatomic, and Angiographic Outcomes with Bioerodible EYP-1901 (vorolanib intravitreal insert) Versus Aflibercept for Diabetic Macular Edema,” Presented at: 49th Macula Society Annual Meeting; San Diego, CA (2026).
- EyePoint, “EyePoint Completes Enrollment of Pivotal Phase 3 Trials for DURAVYU™ in Wet Age-Related Macular Degeneration,” EyePoint, Inc., (2025). Accessed February 20, 2026.
- EyePoint, “EyePoint Reports Corporate Update and Anticipated Pivotal Milestones for 2026,” EyePoint, Inc., (2026). Accessed February 20, 2026.
- EyePoint, “EyePoint Announces Pivotal Phase 3 Program Initiation for DURAVYU™ in Diabetic Macular Edema,” EyePoint, Inc., (2025). Accessed February 20, 2026.
